Back Nitrering Afrikaans نترتة Arabic Nitració Catalan Nitrace Czech Nitrierung German Nitración Spanish Nitreerimine Estonian Nitrazio Basque نیترودار کردن Persian Nitraus Finnish
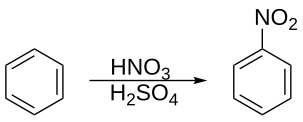
In organic chemistry, nitration is a general class of chemical processes for the introduction of a nitro group (−NO2) into an organic compound. The term also is applied incorrectly to the different process of forming nitrate esters (−ONO2) between alcohols and nitric acid (as occurs in the synthesis of nitroglycerin). The difference between the resulting molecular structures of nitro compounds and nitrates (NO−3) is that the nitrogen atom in nitro compounds is directly bonded to a non-oxygen atom (typically carbon or another nitrogen atom), whereas in nitrate esters (also called organic nitrates), the nitrogen is bonded to an oxygen atom that in turn usually is bonded to a carbon atom (nitrito group).
There are many major industrial applications of nitration in the strict sense; the most important by volume are for the production of nitroaromatic compounds such as nitrobenzene.
Nitration reactions are notably used for the production of explosives, for example the conversion of guanidine to nitroguanidine and the conversion of toluene to trinitrotoluene (TNT). However, they are of wide importance as chemical intermediates and precursors. Millions of tons of nitroaromatics are produced annually.[1]
- ^ Gerald Booth (2007). "Nitro Compounds, Aromatic". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a17_411. ISBN 978-3527306732.
© MMXXIII Rich X Search. We shall prevail. All rights reserved. Rich X Search