
Back توترية Arabic Тоничност Bulgarian Tonija BS Tonicitat Catalan Tonicita Czech Tonizität German Tonicidad (biología) Spanish تونیسیته Persian Tonisitas ID Tonicità Italian
This article needs additional citations for verification. (February 2018) |
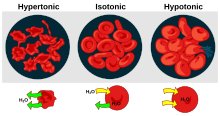

In chemical biology, tonicity is a measure of the effective osmotic pressure gradient; the water potential of two solutions separated by a partially-permeable cell membrane. Tonicity depends on the relative concentration of selective membrane-impermeable solutes across a cell membrane which determine the direction and extent of osmotic flux. It is commonly used when describing the swelling-versus-shrinking response of cells immersed in an external solution.
Unlike osmotic pressure, tonicity is influenced only by solutes that cannot cross the membrane, as only these exert an effective osmotic pressure. Solutes able to freely cross the membrane do not affect tonicity because they will always equilibrate with equal concentrations on both sides of the membrane without net solvent movement. It is also a factor affecting imbibition.
There are three classifications of tonicity that one solution can have relative to another: hypertonic, hypotonic, and isotonic.[1] A hypotonic solution example is distilled water.
- ^ Sperelakis, Nicholas (2011). Cell Physiology Source Book: Essentials of Membrane Biophysics. Academic Press. p. 288. ISBN 978-0-12-387738-3.
© MMXXIII Rich X Search. We shall prevail. All rights reserved. Rich X Search