
Back Stabiele isotoop Afrikaans نظير مستقر Arabic Isótopu estable AST Стабилен изотоп Bulgarian Isotop#Stabile Isotope German Ευσταθή νουκλίδια Greek Stabila izotopo Esperanto Isótopo estable Spanish Stabiilne isotoop Estonian ایزوتوپ پایدار Persian
This article needs additional citations for verification. (December 2018) |
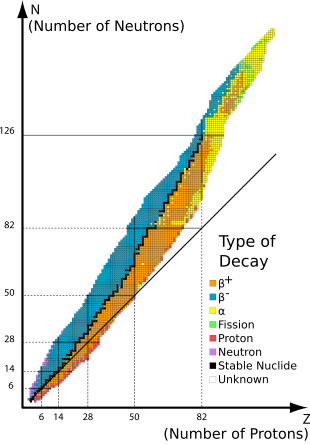
Stable nuclides are nuclides that are not radioactive and so (unlike radionuclides) do not spontaneously undergo radioactive decay.[1] When such nuclides are referred to in relation to specific elements, they are usually termed stable isotopes.
The 80 elements with one or more stable isotopes comprise a total of 251 nuclides that have not been known to decay using current equipment (see list at the end of this article). Of these 80 elements, 26 have only one stable isotope; they are thus termed monoisotopic. The rest have more than one stable isotope. Tin has ten stable isotopes, the largest number of stable isotopes known for an element.
- ^ "DOE explains ... Isotopes". Department of Energy, United States. Archived from the original on 14 April 2022. Retrieved 11 January 2023.
© MMXXIII Rich X Search. We shall prevail. All rights reserved. Rich X Search