
Back Yster(III)oksied Afrikaans أكسيد الحديد الثلاثي Arabic Óxidu de fierro (III) AST Ferritlər Azerbaijani اوکسید دمیر (III) AZB Дижелезен триоксид Bulgarian ফেরিক অক্সাইড Bengali/Bangla Òxid de ferro(III) Catalan Oxid železitý Czech Eisen(III)-oxid German
 | |

| |
| |
| Names | |
|---|---|
| IUPAC name
Iron(III) oxide
| |
| Other names | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.013.790 |
| EC Number |
|
| E number | E172(ii) (colours) |
| 11092 | |
| KEGG | |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| Properties | |
| Fe2O3 | |
| Molar mass | 159.687 g·mol−1 |
| Appearance | Red solid |
| Odor | Odorless |
| Density | 5.25 g/cm3[1] |
| Melting point | 1,539 °C (2,802 °F; 1,812 K)[1] decomposes 105 °C (221 °F; 378 K) β-dihydrate, decomposes 150 °C (302 °F; 423 K) β-monohydrate, decomposes 50 °C (122 °F; 323 K) α-dihydrate, decomposes 92 °C (198 °F; 365 K) α-monohydrate, decomposes[2] |
| Insoluble | |
| Solubility | Soluble in diluted acids,[1] barely soluble in sugar solution[2] Trihydrate slightly soluble in aq. tartaric acid, citric acid, CH3COOH[2] |
| +3586.0x10−6 cm3/mol | |
Refractive index (nD)
|
n1 = 2.91, n2 = 3.19 (α, hematite)[3] |
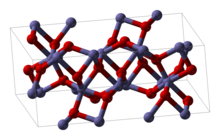
| Structure | |
| Rhombohedral, hR30 (α-form)[4] Cubic bixbyite, cI80 (β-form) Cubic spinel (γ-form) Orthorhombic (ε-form)[5] | |
| R3c, No. 161 (α-form)[4] Ia3, No. 206 (β-form) Pna21, No. 33 (ε-form)[5] | |
| 3m (α-form)[4] 2/m 3 (β-form) mm2 (ε-form)[5] | |
| Octahedral (Fe3+, α-form, β-form)[4] | |
| Thermochemistry[6] | |
Heat capacity (C)
|
103.9 J/mol·K[6] |
Std molar
entropy (S⦵298) |
87.4 J/mol·K[6] |
Std enthalpy of
formation (ΔfH⦵298) |
−824.2 kJ/mol[6] |
Gibbs free energy (ΔfG⦵)
|
−742.2 kJ/mol[6] |
| Hazards | |
| GHS labelling: | |
 [7] [7]
| |
| Warning | |
| H315, H319, H335[7] | |
| P261, P305+P351+P338[7] | |
| NFPA 704 (fire diamond) | |
Threshold limit value (TLV)
|
5 mg/m3[1] (TWA) |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
10 g/kg (rats, oral)[9] |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
TWA 10 mg/m3[8] |
REL (Recommended)
|
TWA 5 mg/m3[8] |
IDLH (Immediate danger)
|
2500 mg/m3[8] |
| Related compounds | |
Other anions
|
Iron(III) fluoride |
Other cations
|
Manganese(III) oxide Cobalt(III) oxide |
Related iron oxides
|
Iron(II) oxide Iron(II,III) oxide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |

Iron(III) oxide or ferric oxide is the inorganic compound with the formula Fe2O3. It is one of the three main oxides of iron, the other two being iron(II) oxide (FeO), which is rare; and iron(II,III) oxide (Fe3O4), which also occurs naturally as the mineral magnetite. As the mineral known as hematite, Fe2O3 is the main source of iron for the steel industry. Fe2O3 is readily attacked by acids. Iron(III) oxide is often called rust, since rust shares several properties and has a similar composition; however, in chemistry, rust is considered an ill-defined material, described as hydrous ferric oxide.[10]
- ^ a b c d Haynes, p. 4.69
- ^ a b c Comey, Arthur Messinger; Hahn, Dorothy A. (February 1921). A Dictionary of Chemical Solubilities: Inorganic (2nd ed.). New York: The MacMillan Company. p. 433.
- ^ Haynes, p. 4.141
- ^ a b c d Ling, Yichuan; Wheeler, Damon A.; Zhang, Jin Zhong; Li, Yat (2013). Zhai, Tianyou; Yao, Jiannian (eds.). One-Dimensional Nanostructures: Principles and Applications. Hoboken, New Jersey: John Wiley & Sons, Inc. p. 167. ISBN 978-1-118-07191-5.
- ^ a b c Cite error: The named reference
atmilabwas invoked but never defined (see the help page). - ^ a b c d e Haynes, p. 5.12
- ^ a b c Sigma-Aldrich Co., Iron(III) oxide. Retrieved on 2014-07-12.
- ^ a b c NIOSH Pocket Guide to Chemical Hazards. "#0344". National Institute for Occupational Safety and Health (NIOSH).
- ^ a b "SDS of Iron(III) oxide" (PDF). KJLC. England: Kurt J Lesker Company Ltd. 5 January 2012. Retrieved 12 July 2014.
- ^ PubChem. "Iron oxide (Fe2O3), hydrate". pubchem.ncbi.nlm.nih.gov. Retrieved 11 November 2020.
© MMXXIII Rich X Search. We shall prevail. All rights reserved. Rich X Search
