
Back نظام أصهري Arabic Evtektika Azerbaijani Эўтэктыка Byelorussian ইউটেকটিক ব্যবস্থা Bengali/Bangla Eutektička tačka BS Mescla eutèctica Catalan Eutektikum Czech Eutektikum German Ευτηκτικό σύστημα Greek Eŭtekto Esperanto
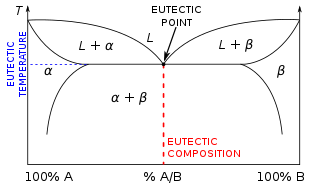
A eutectic system or eutectic mixture (/juːˈtɛktɪk/ yoo-TEK-tik)[1] is a homogeneous mixture that has a melting point lower than those of the constituents.[2] The lowest possible melting point over all of the mixing ratios of the constituents is called the eutectic temperature. On a phase diagram, the eutectic temperature is seen as the eutectic point (see plot on the right).[3]
Non-eutectic mixture ratios have different melting temperatures for their different constituents, since one component's lattice will melt at a lower temperature than the other's. Conversely, as a non-eutectic mixture cools down, each of its components solidifies into a lattice at a different temperature, until the entire mass is solid.
Not all binary alloys have eutectic points, since the valence electrons of the component species are not always compatible[clarification needed] in any mixing ratio to form a new type of joint crystal lattice. For example, in the silver-gold system the melt temperature (liquidus) and freeze temperature (solidus) "meet at the pure element endpoints of the atomic ratio axis while slightly separating in the mixture region of this axis".[4]
In the real world, eutectic properties can be used to advantage in such processes as eutectic bonding, where silicon chips are bonded to gold-plated substrates with ultrasound, and eutectic alloys prove valuable in such diverse applications as soldering, brazing, metal casting, electrical protection, fire sprinkler systems, and nontoxic mercury substitutes. By managing phase transformation during solidification, a suitable eutectic alloy can be made stronger than any of its individual components, a valuable property in an extreme application such as the hypereutectic cast aluminum pistons used in the high-revving 550 hp (410 kW) twin-turbo intercooled DOHC Cadillac Blackwing V8 introduced in 2018.
The term eutectic was coined in 1884 by British physicist and chemist Frederick Guthrie (1833–1886). The word originates from Greek εὐ- (eû) 'well', and τῆξῐς (têxis) 'melting'.[2]
- ^ "eutectic". Merriam-Webster.com Dictionary.
- ^ a b Guthrie, Frederick (June 1884). "LII. On eutexia". The London, Edinburgh, and Dublin Philosophical Magazine and Journal of Science. 5th series. 17 (108): 462–482. doi:10.1080/14786448408627543. p. 462:
The main argument of the present communication hinges upon the existence of compound bodies, whose chief characteristic is the lowness of their temperatures of fusion. This property of the bodies may be called Eutexia, the bodies possessing it eutectic bodies or eutectics (εὖ τήκειν).
- ^ Smith & Hashemi 2006, pp. 326–327
- ^ "Collection of Phase Diagrams". www.crct.polymtl.ca.
© MMXXIII Rich X Search. We shall prevail. All rights reserved. Rich X Search